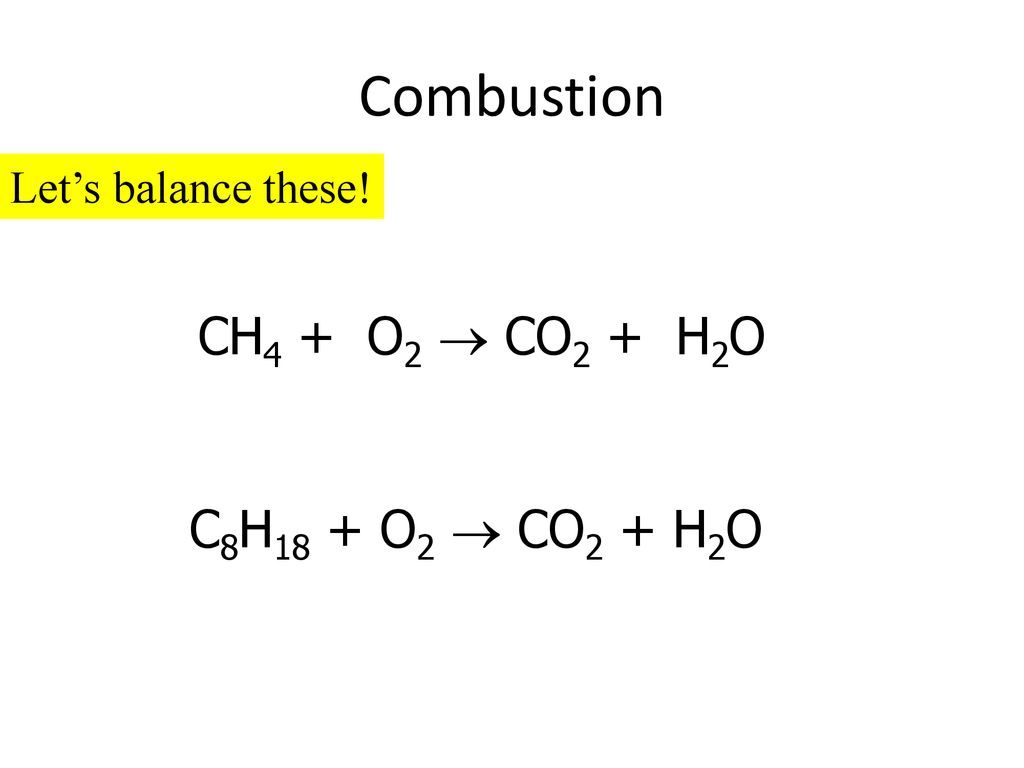
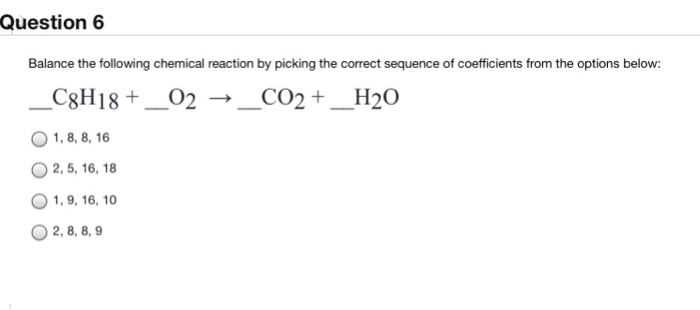
Balance the following chemical equation, then answer the following question. C8H18(g) + O2(g) - Brainly.com

Determine the balanced chemical equation for this reaction. C8H18(g)+O2(g)→ CO2(g)+H2O(g) - Home Work Help - Learn CBSE Forum
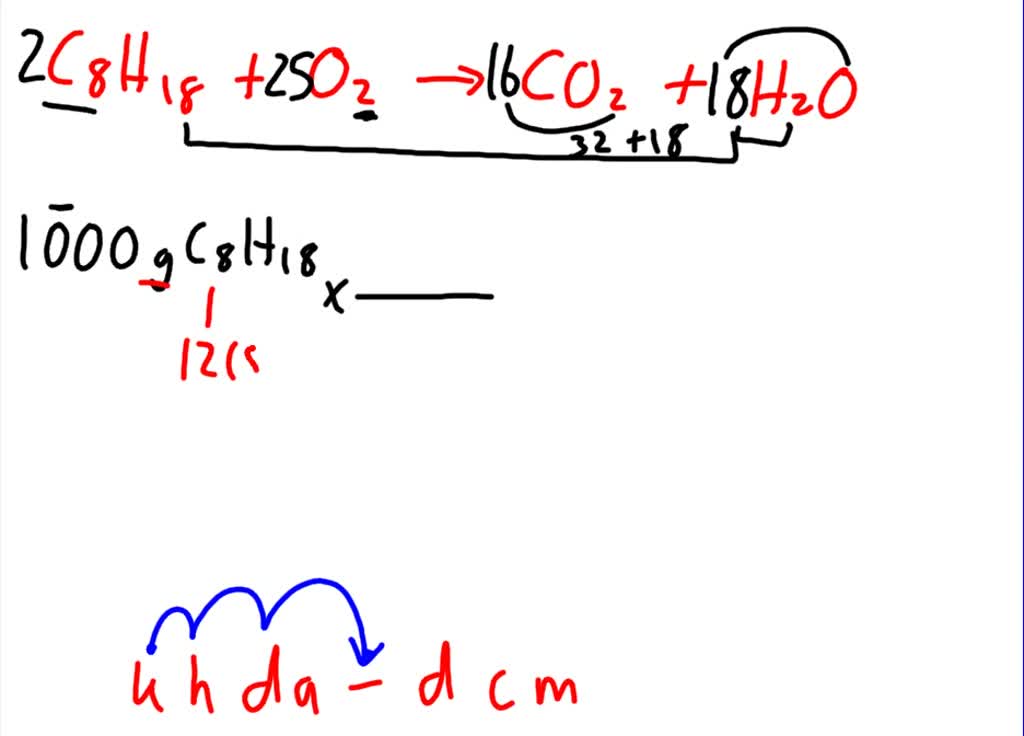
SOLVED:The combustion of gasoline produces carbon dioxide and water. Assume gasoline to be pure octane (C8H18) and calculate the mass (in kg) of carbon dioxide that is added to the atmosphere per

SOLVED:Give the balanced equation for each of the following. a, The combustion of ethanol (C2 H3 OH) forms carbon dioxide and water vapor. A combustion reaction refers to a reaction of a